|
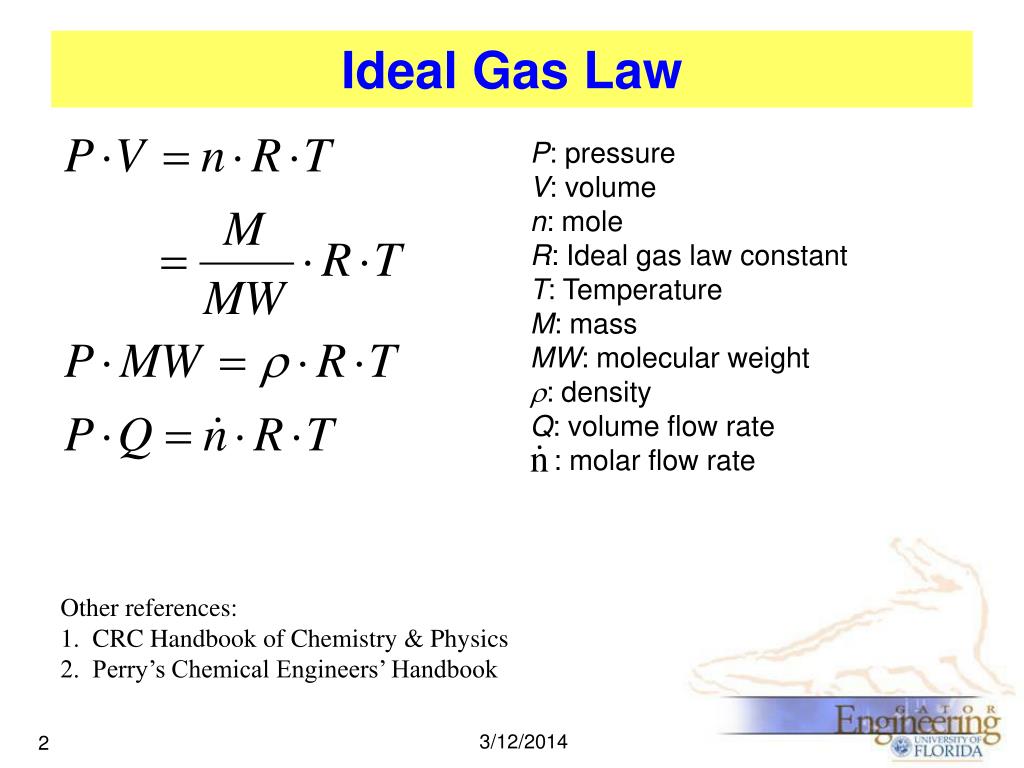
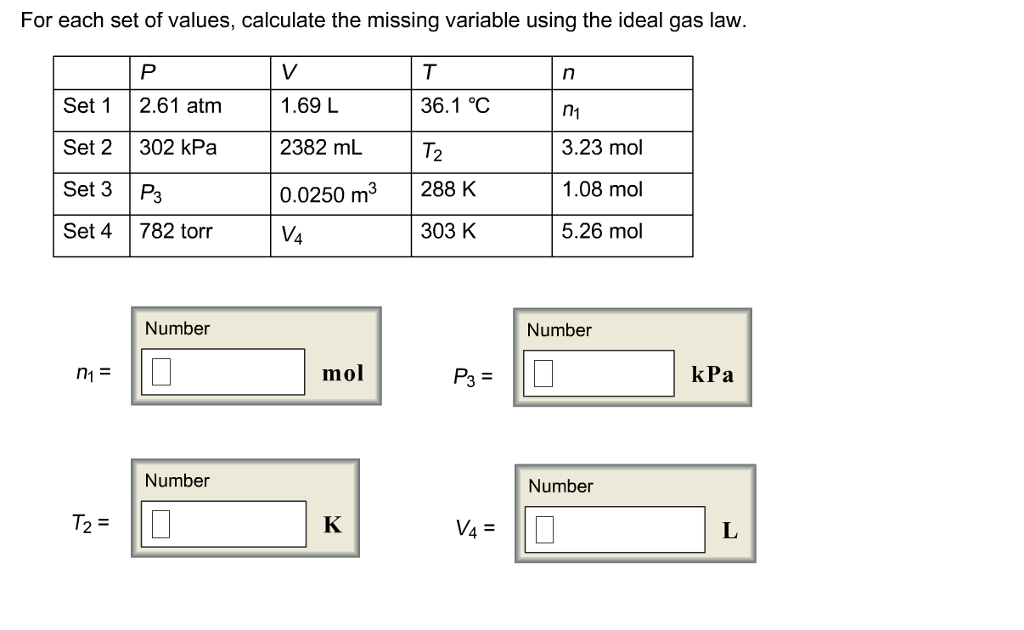
The law has a simple mathematical form if the temperature is measured on an absolute scale, such as in kelvins. Pressure and temperature will both increase or decrease simultaneously as long as the volume is held constant. Simplified, this means that if you increase the temperature of a gas, the pressure rises proportionally. In other words, Gay-Lussac's Law states that the pressure of a fixed amount of gas at fixed volume is directly proportional to its temperature in kelvins. Gay-Lussac’s Law is an ideal gas law where at constant volume, the pressure of an ideal gas is directly proportional to its absolute temperature. If we add 0.250 mol of gas at the same pressure and temperature, what is the final total volume of the gas? Thus, its molar volume at STP is 22.71 LĪ 6.00 L sample at 25.0 ☌ and 2.00 atm contains 0.500 mol of gas. One mole of an ideal gas occupies 22.71 L at STP. #V/n = k#, where #k# is a proportionality constant.Įqual volumes of hydrogen, oxygen, or carbon dioxide contain the same number of molecules. #V ∝ n#, where #V# is the volume, and #n# is the number of moles. It does not depend on the sizes or the masses of the molecules. The volume increases as the number of moles increases. Public License: CC Attribution-ShareAlike 4.Another statement is, "Volume is directly proportional to the number of moles." Compressibility factor Source: Wikipedia URL: 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
